Staphylococcus aureus
Staphylococcus aureus can infect every niche of the human host and is the leading cause of Gram-positive sepsis. In the United States, there are over 900,000 severe S. aureus infections annually of which nearly 10 % are caused by strains that are resistant to commonly used antibiotics. Both the CDC and WHO have identified S. aureus as a pathogen that necessitates the development of more effective treatments. We use a combination of analytical chemistry, bacterial pathogenesis, bacteriology, and chemical biology techniques to better understand how the vertebrate host kills S. aureus and how S. aureus evades killing by the host. We identify S. aureus pathways targeted by aberrant post-translational modifications at the host-pathogen interface. These pathways represent potential therapeutic weaknesses in S. aureus and following validation they will be targeted with novel antimicrobial compounds that sensitize S. aureus to killing by the host’s immune system.
Polyunsaturated Fatty Acids
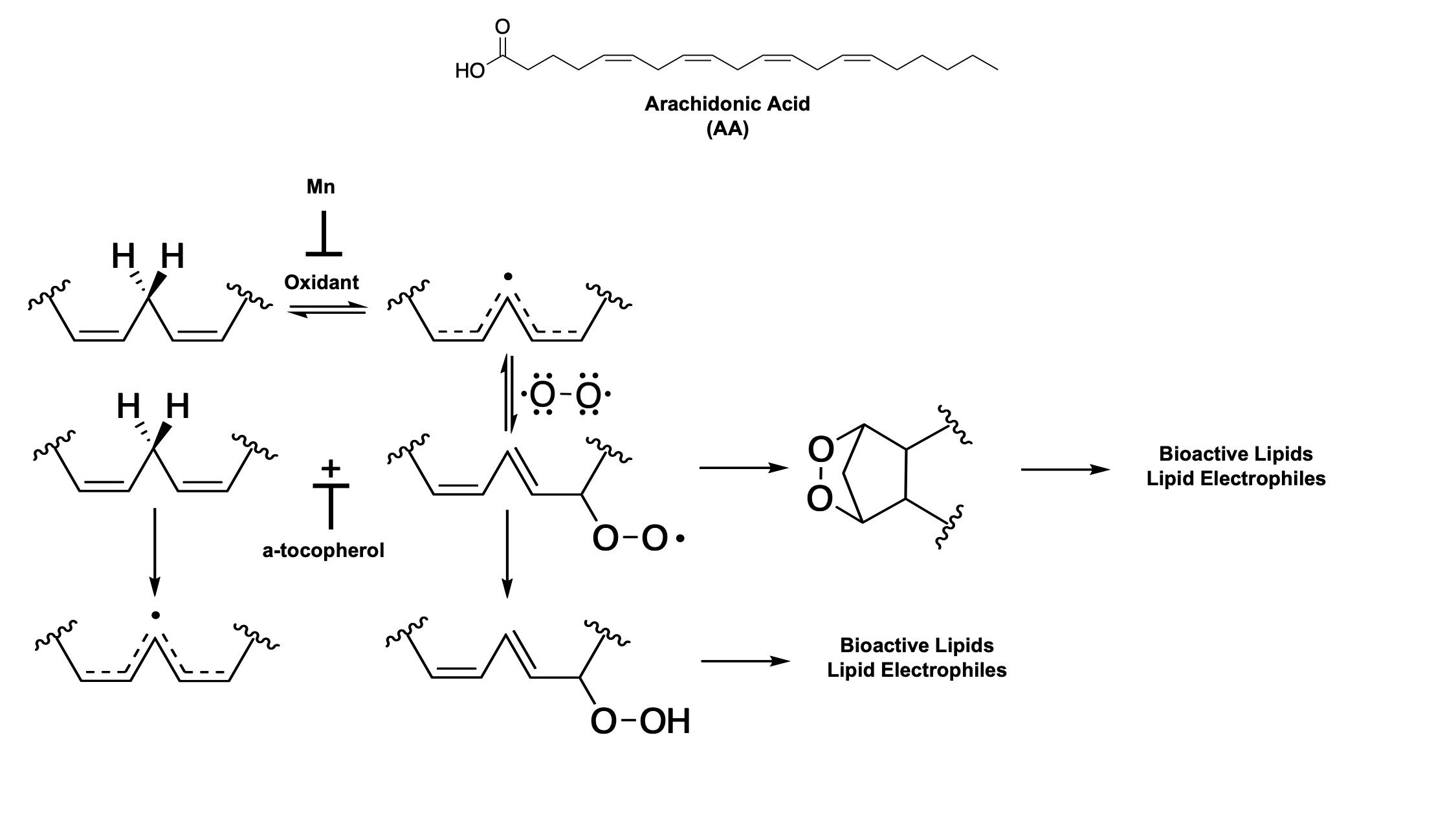
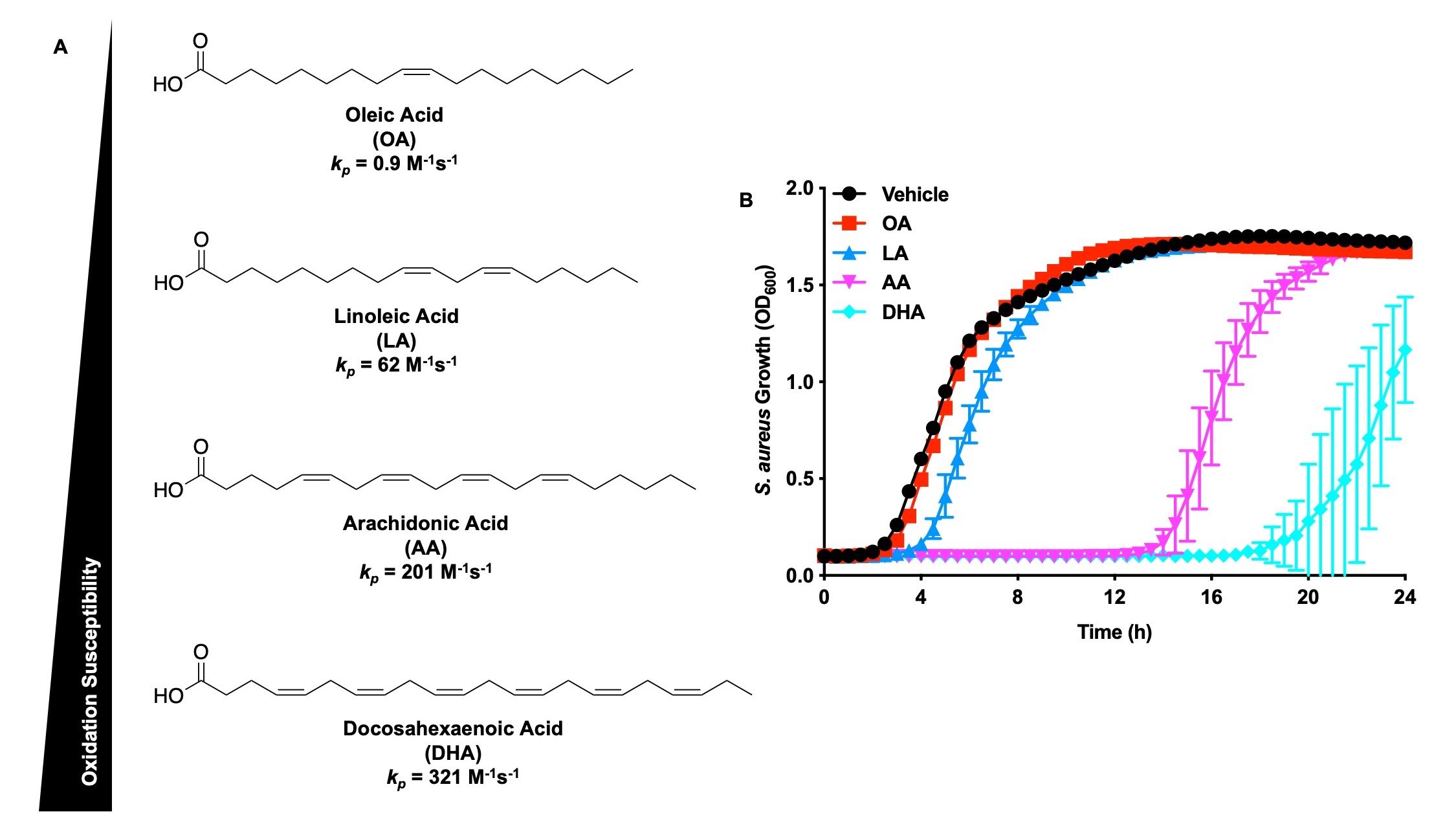
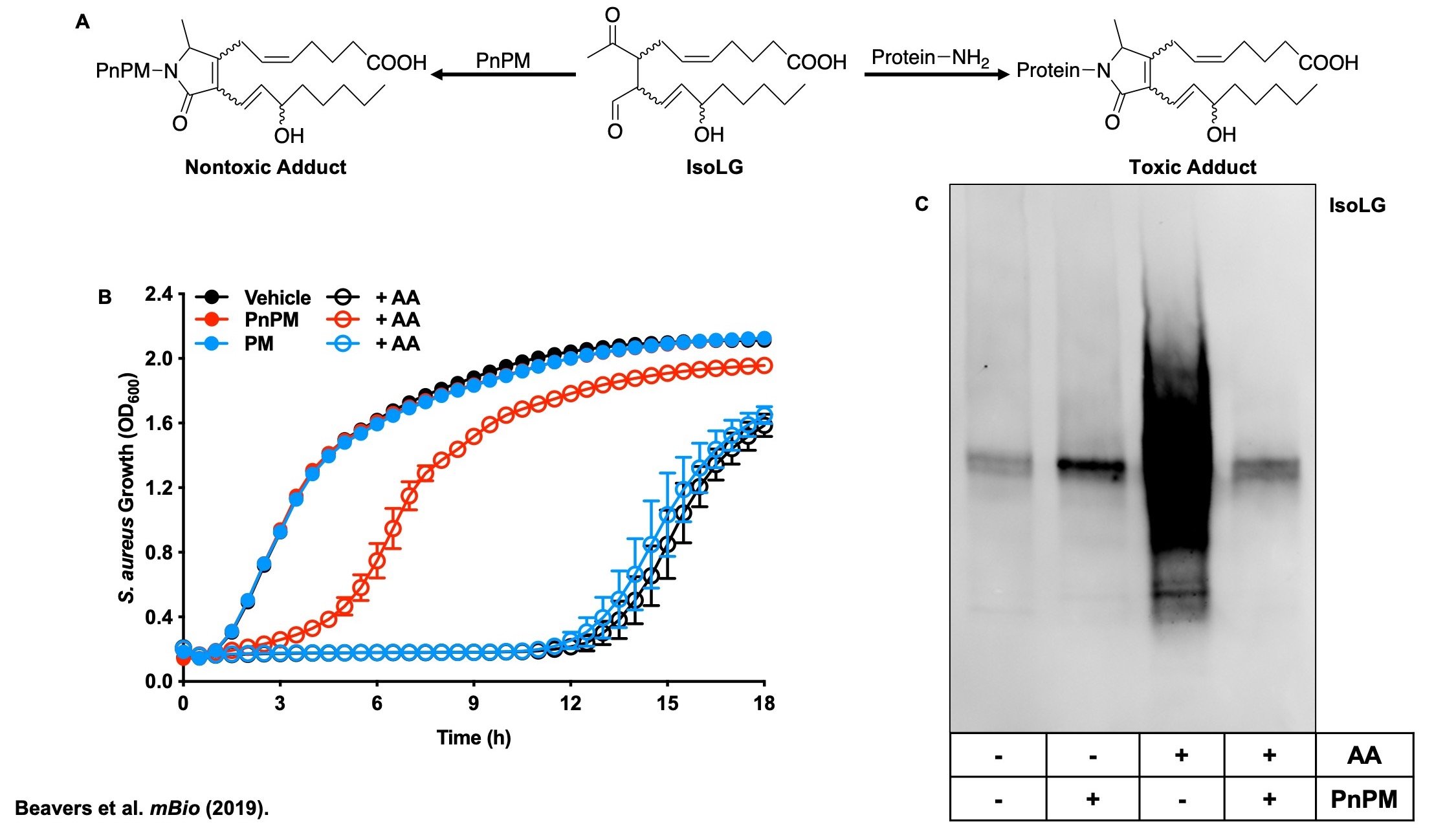
When immune cells of the vertebrate host encounter S. aureus they release polyunsaturated fatty acids (PUFA), which are bactericidal against S. aureus. PUFAs are incorporated into S. aureus phospholipids where they are oxidized to various lipid electrophiles. Nucleophilic functional groups of S. aureus macromolecules react with these lipid electrophiles with potentially deleterious effects. We are interested in defining the role of lipid electrophile post-translational modifications at the host-pathogen interface. Active projects in the group seek to discover the identity of the electrophiles, the S. aureus pathways responsible for electrophile generation, and the S. aureus proteins targeted by electrophiles. Finally, we aim to define the role of lipid electrophiles in S. aureus pathogenesis, and determine if this strategy can be used as an effective antimicrobial therapy.
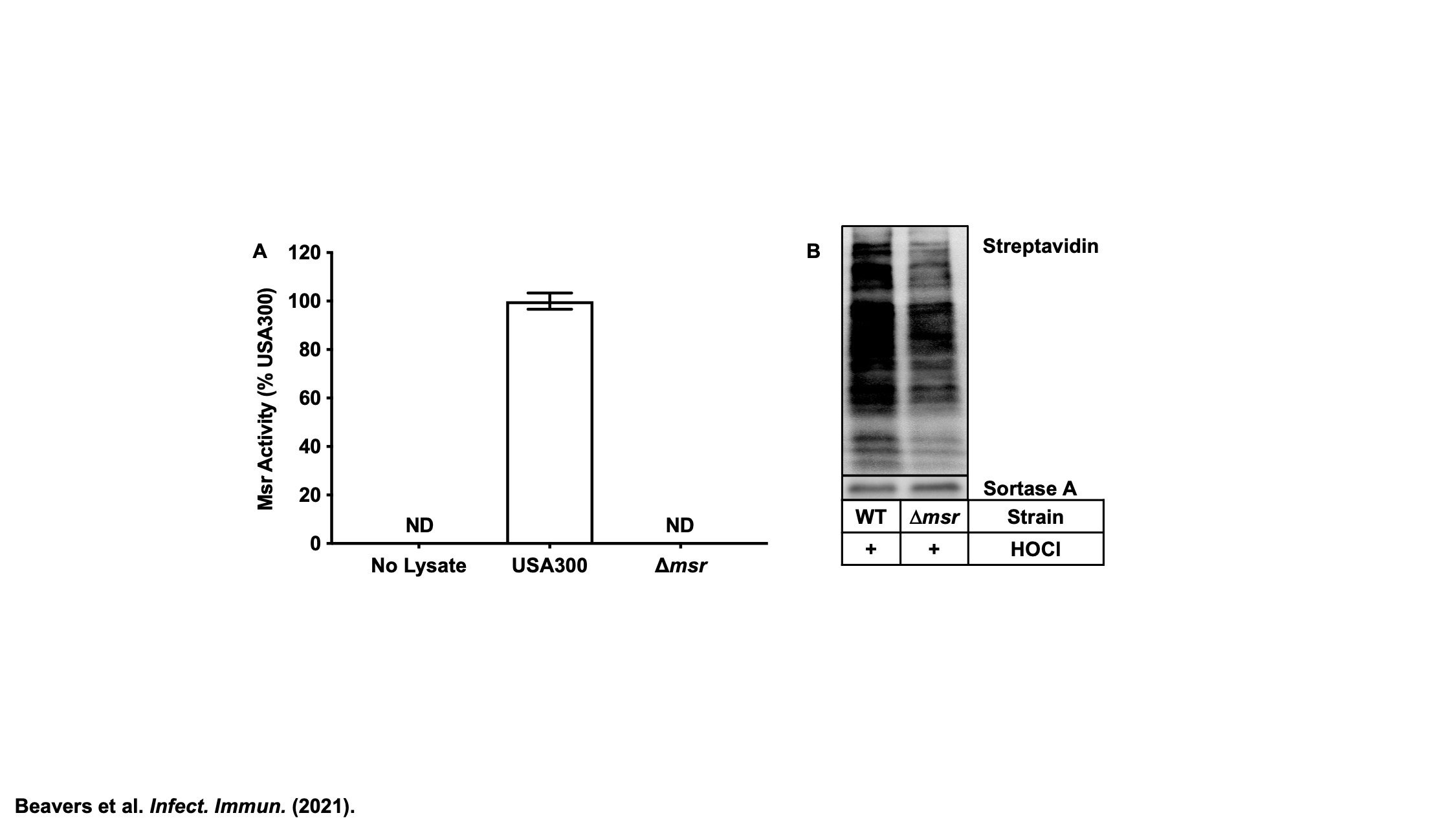
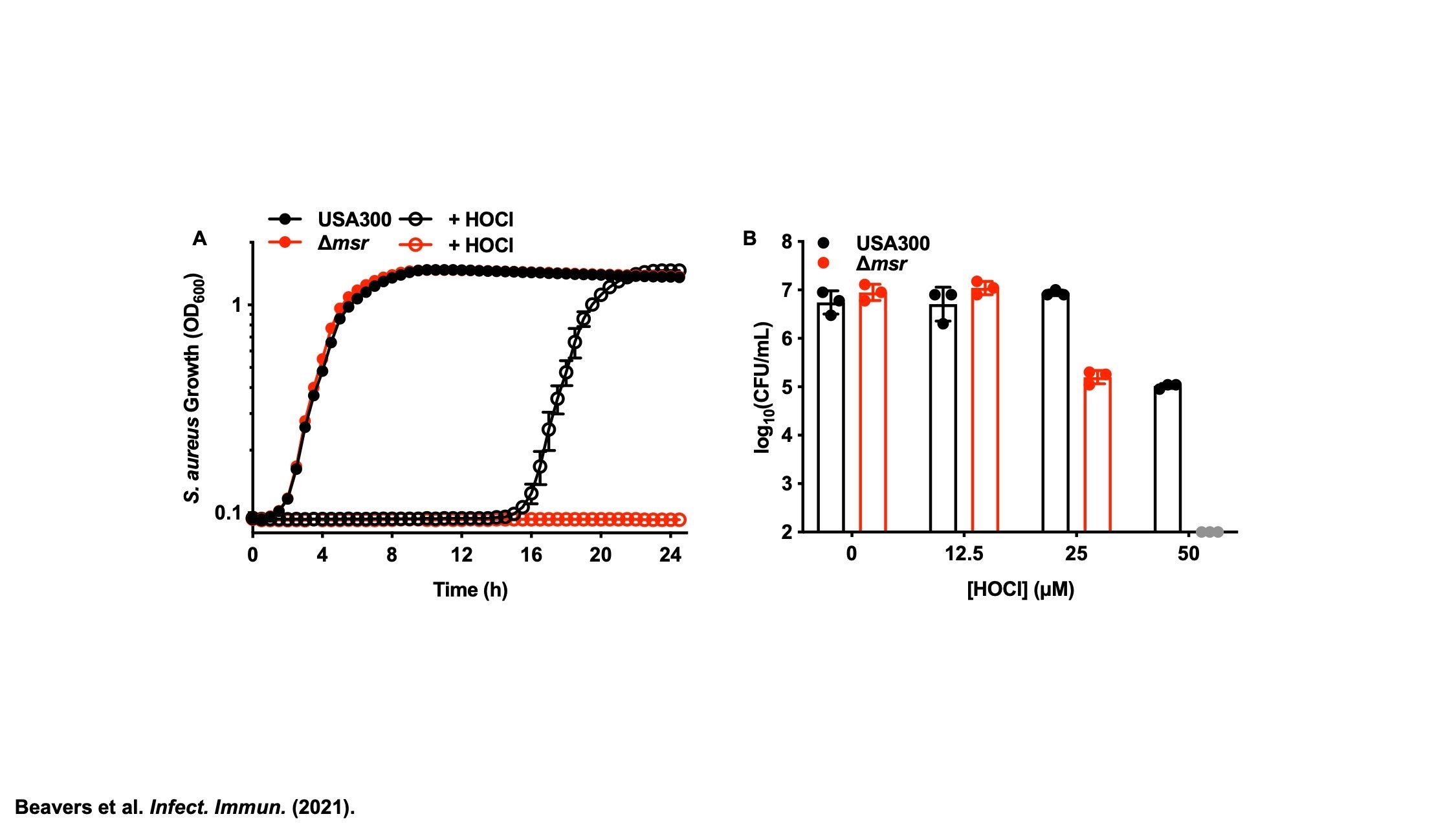
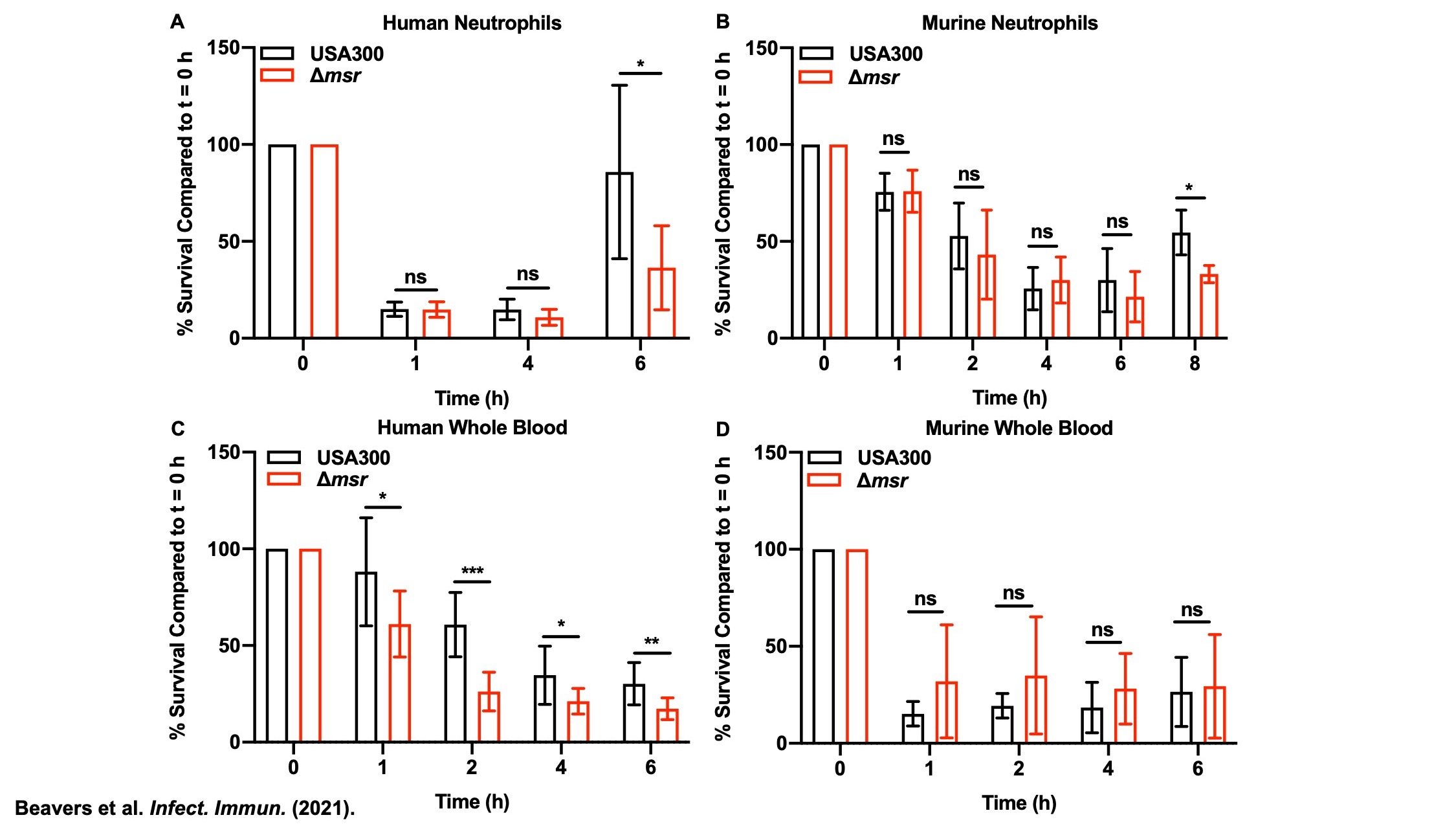
Methionine Sulfoxide Reductases
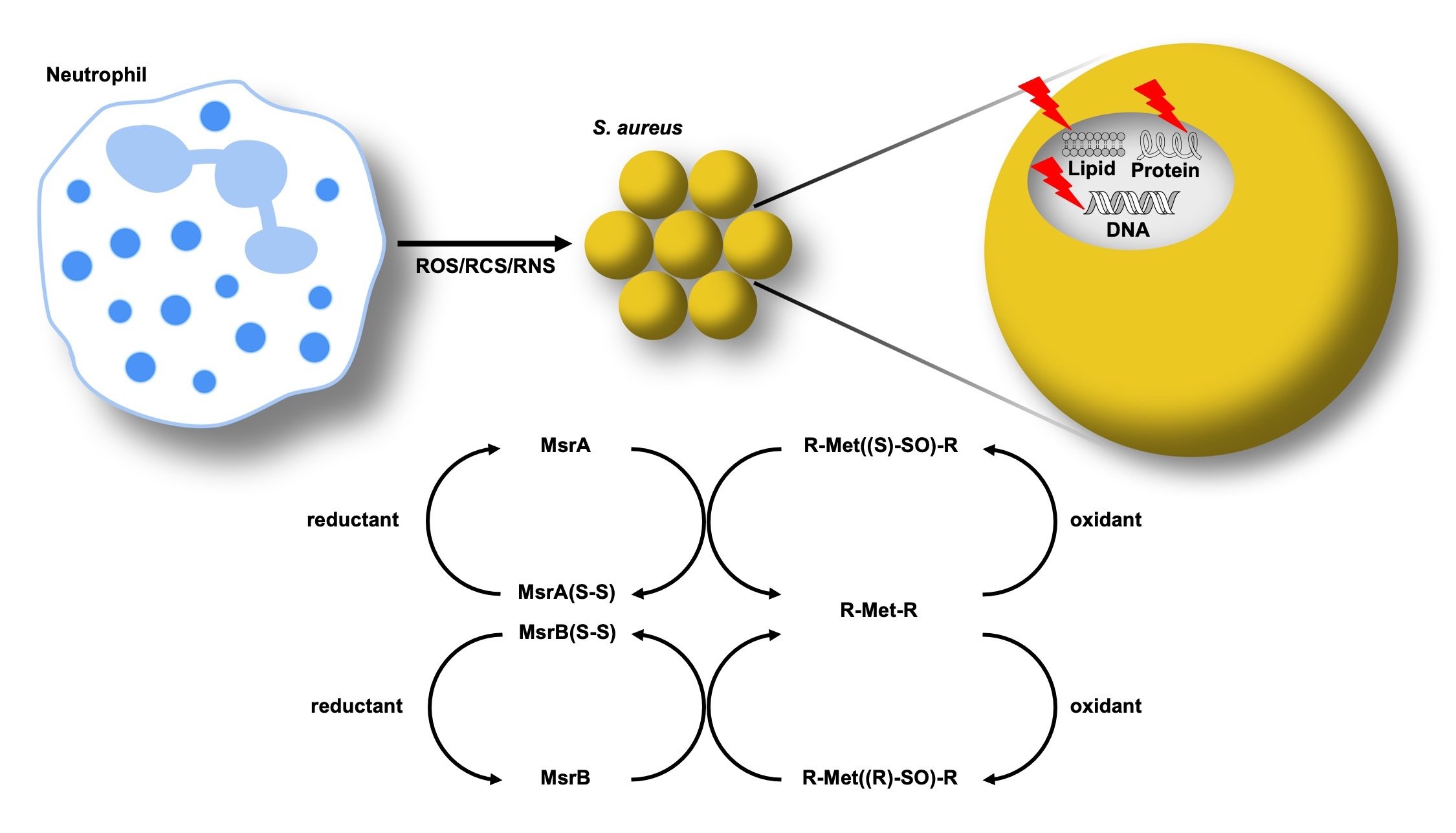
The sulfur containing amino acids, cysteine and methionine, are susceptible to oxidation. Unlike cysteine, the sulfur atom of methionine does not have a direct role in catalysis, but contributes to protein stability through hydrophobic and sulfur-π interactions with other amino acids. Therefore, methionine oxidation can have deleterious effects on S. aureus enzymes. The methionine sulfoxide reductases (Msr) evolved to repair oxidized methionine (methionine sulfoxide), preventing the need to resynthesize oxidized proteins de novo. Staphylococci are unique in having four, non-redundant Msr enzymes. Active projects in the group seek to identify the substrate specificity of each Msr, define the regulation of each Msr, and determine the role of each Msr in S. aureus pathogenesis. Understanding how S. aureus uses these enzymes to subvert host antimicrobial strategies will inform future antimicrobial design to render S. aureus more vulnerable to oxidative killing.
LSU-SVM Mass Spectrometry Resource Center
The LSU SVM Mass Spectrometry Resource Center, which houses a state-of-the-art mass spectrometry system: the Shimadzu 8060NX triple quadrupole mass spectrometer with a Shimadzu Nexera XS 40 series UHPLC, is now accepting samples. This instrumentation is capable of performing small molecule identification and quantification, lipidomics, and metabolomics at femotmole levels from complex biological matrices. Please contact wbeavers@lsu.edu with any project inquiries.